
Population Pharmacokinetics Ppt Video Online Download
Pharmacokinetics (PK) studies are of great importance in creating new therapeutic drugs, understanding both the beneficial and harmful effects of drugs, and guiding drug development experiments and trials In PK assays, the applied assay method should be well characterized and fully validated, in order to yield reliable resultsObtained providing a robust assessment of the PK endpoints PK parameters are all derived from the concentration information obtained from samples assayed by the bioanalytical laboratory Therefore, understanding the limits of detection of the bioanalytical methods for the analytes and metabolites is an important consideration in PK study
Pk assay fda guidance
Pk assay fda guidance-Active drug assays can be validated according to current pk assay validation guidelines (ema, fda, ) for some aspects however, the guidelines do not consider special needs of active drug assays for validation and sample analysis ø general validation concept ø analytical run acceptance criteria ø investigation of matrix effect and conclusions øBinding or LC–MS technology, and the assays are validated and applied following the guidelines from various regulatory agencies in their countries (eg, US FDA, EMEA or Health Canada) 1 These bioanalytical method guidelines focus on the PK of traditional drug compounds;

Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis
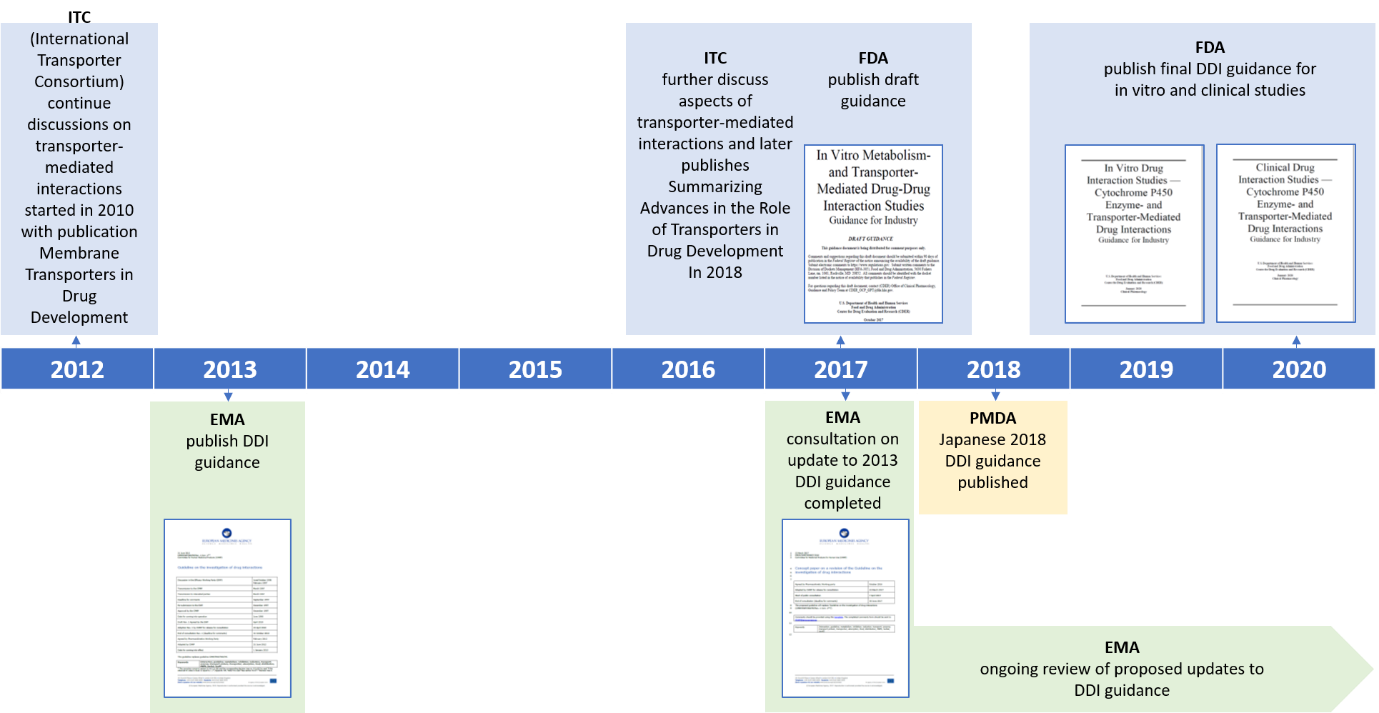
FDA Drugdrug Interaction Guidance A Comparison Analysis and Action Plan by Pharmaceutical Industrial Scientists The new clinical DDI guidance from the FDA now has even higher harmonization with the guidance (or guidelines) from the EMA and PMDA• BAV Guidelines (three topics)A typical suite of assays required during biotherapeutic development includes drug concentration assay (pharmacokinetic or PK assay) and assays detecting presence of antidrug immune response Multiple assays may be required to determine appropriate compound PK as well as to characterize an antidrug immune response
In 1991, the Food and Drug Administration (FDA) made new guidance available for bioanalytical method validation, which informed all laboratories doing bioanalysis how they should validate their scientific methods, its central focus being on methods for the evaluation of Pharmacokinetics (PK) – Guidance updated 01 & 13 (draft)Excellent PK Testing Studies, PK Assay Sample Analysis, And SAD MAD Pharmacokinetics Study Services For Your Drug And Metabolite Bioanalysis sales@nebiolabcom Email Your Inquiry (3) 361 3780 Speak to Our ScientistsTion of a drug's human PK early in drug discovery is important A misprediction of human PK at the preclinical stage, however, is less likely to lead to safety concerns in the clinic This is because the doses chosen are low and can be readily adjusted based on the resultant PK data in the singleascendingdose (SAD) phase in the
Pk assay fda guidanceのギャラリー
各画像をクリックすると、ダウンロードまたは拡大表示できます
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
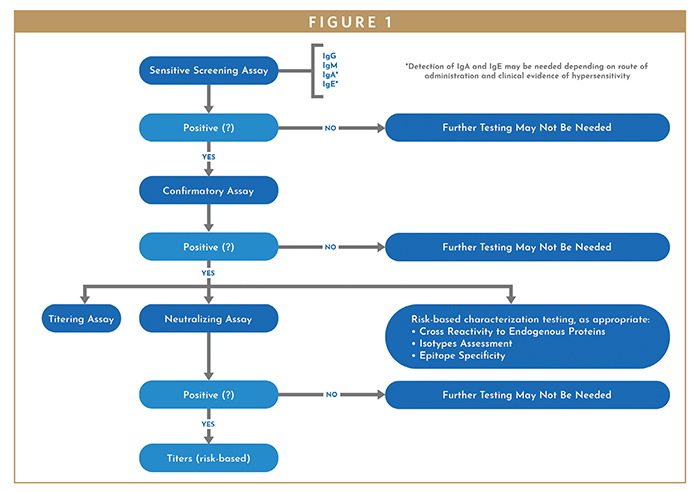 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | 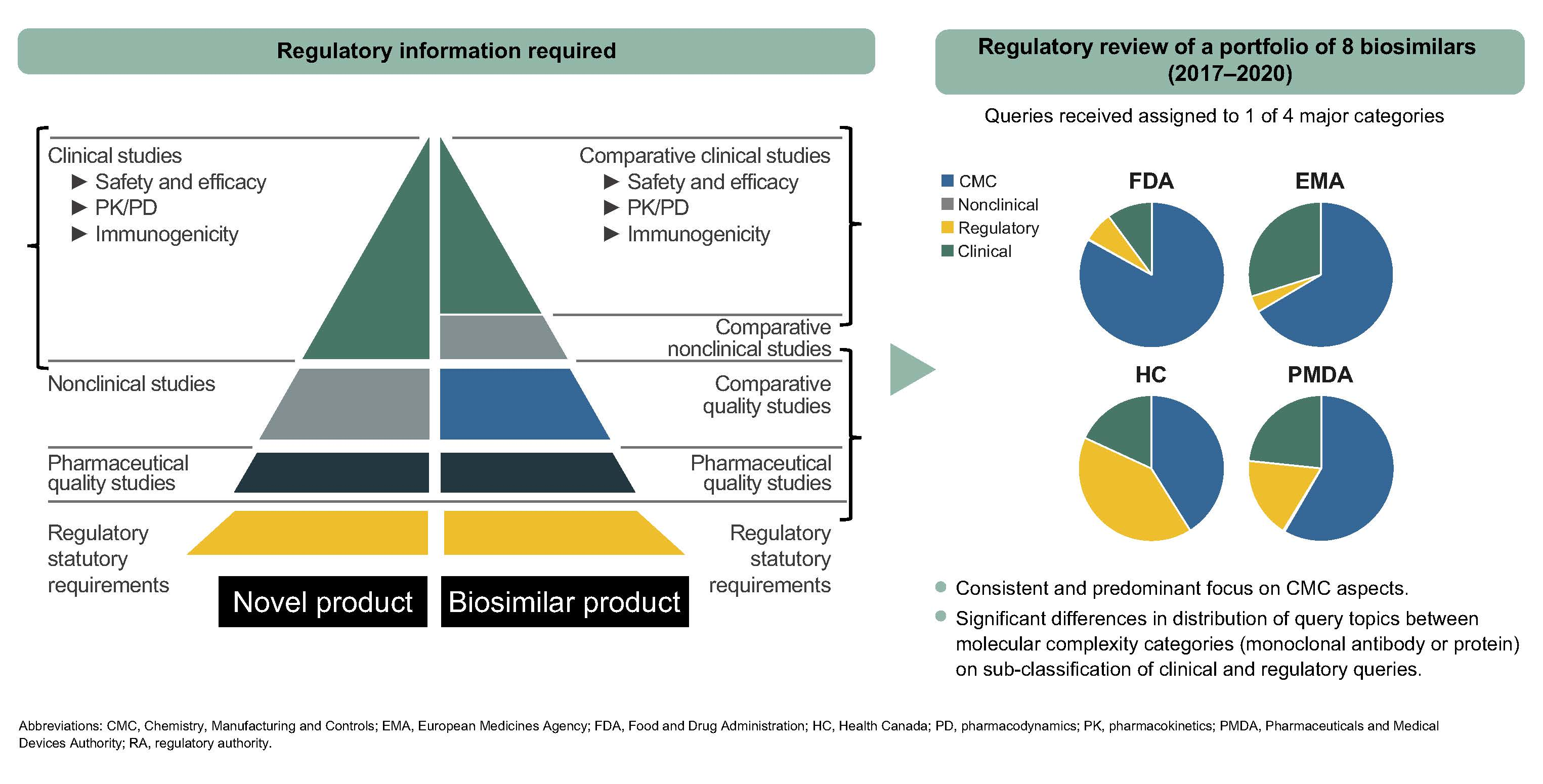 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | 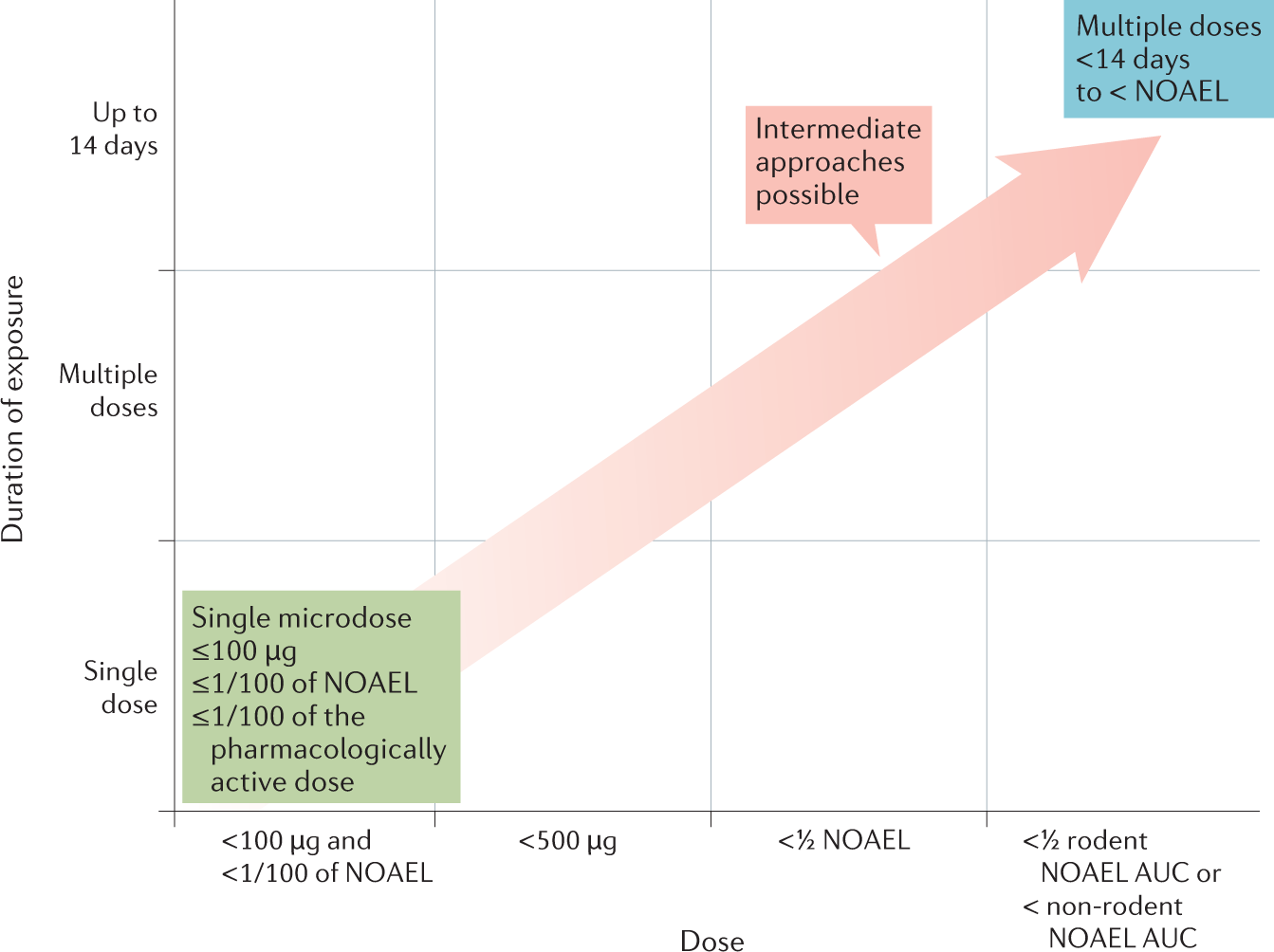 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
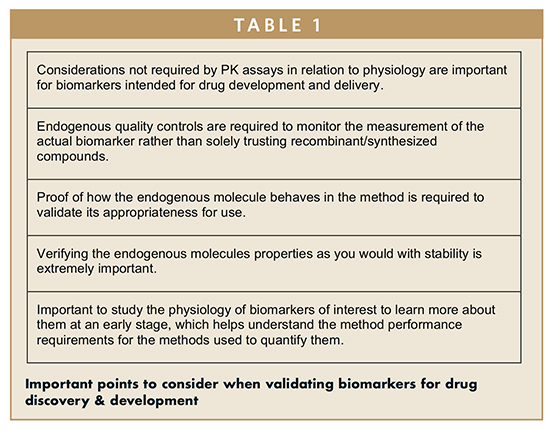 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis | Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
「Pk assay fda guidance」の画像ギャラリー、詳細は各画像をクリックしてください。
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
 Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |  Recommendations For Adaptation And Validation Of Commercial Kits For Biomarker Quantification In Drug Development Bioanalysis |
According to the FDA Guidance for determining the LLOQ, the lowest standard should be selected with an analyte response which is at least 5 times the blank response, has precision of % or less, and allows accuracy of 80–1% Here 5 ng/ml would be designated as the LLOQ• FDA recommends interassay precision be assessed on at least 3 different days with two analysts each preparing a minimum of 6 independent preparations of the sample using the same instrument platform and model • IntraAssay Precision six independent preparations on a single plate • FDA recommends a coefficient of variance (%CV) to
Incoming Term: pk assay fda guidance,




0 件のコメント:
コメントを投稿